
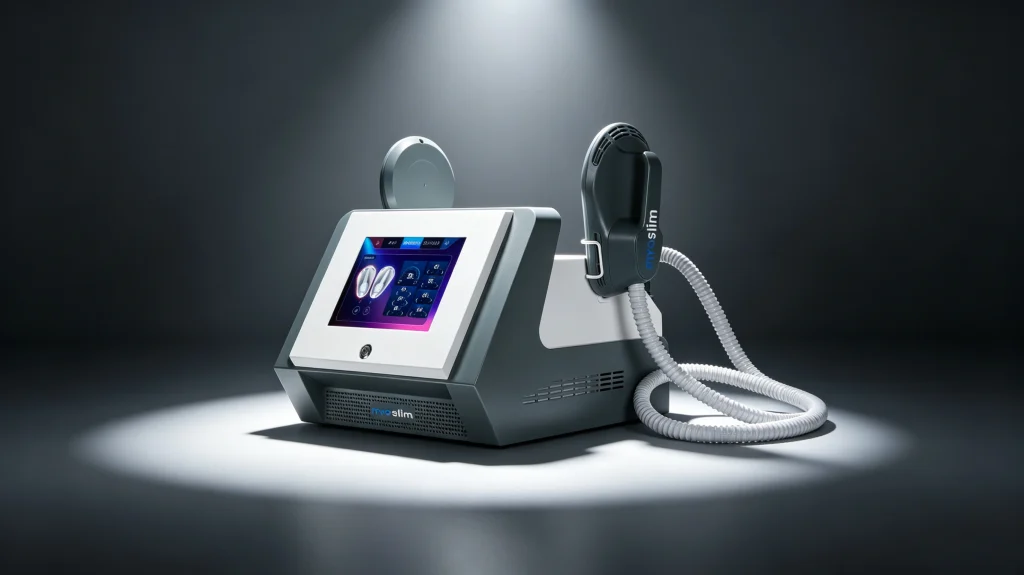
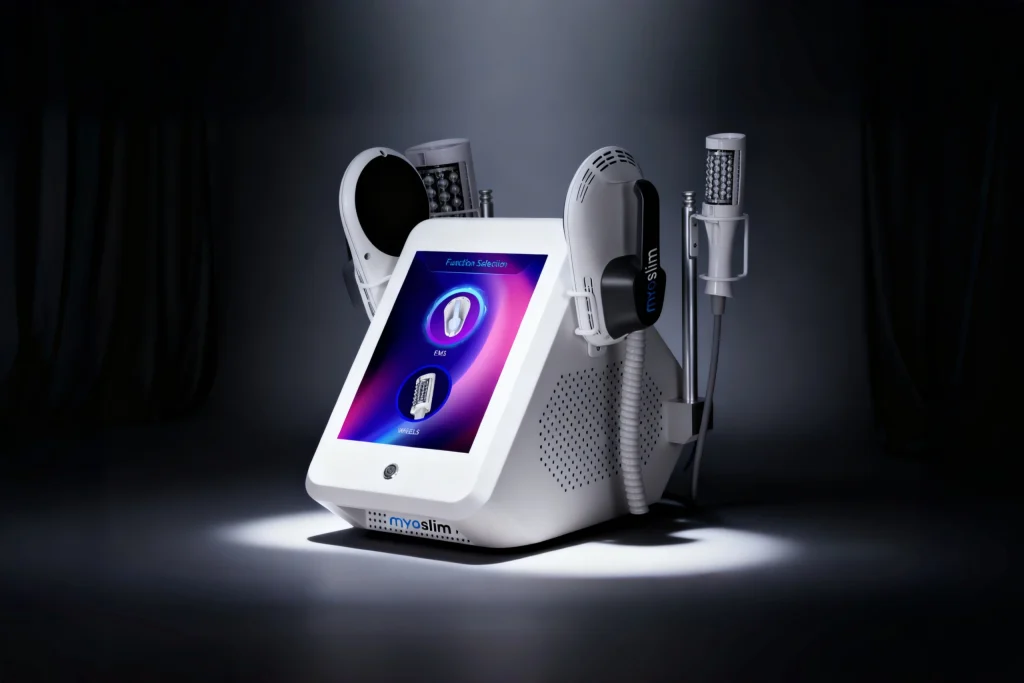
MYOslim Quad White
-8%$2,999.00 Original price was: $2,999.00.$2,749.00Current price is: $2,749.00.


MYOslim Quad White
-8%$2,999.00 Original price was: $2,999.00.$2,749.00Current price is: $2,749.00.
HI-EMT body sculpting system delivering supramaximal muscle contractions, fat reduction, and skin tightening for abdomen, buttocks, arms, and thighs.
Medical Aesthetics · HI-EMT Series
MYOslim Quad White
EMS Body Sculpt series integrates HI-EMT Technology (High‑Intensity Focused Electromagnetic Waves) to induce supramaximal muscle contractions — stimulating deep muscle growth, remodeling internal structure, and promoting simultaneous fatty acid release from triglycerides. The result: visible toning, firming, and reshaping without surgery.
8″ Touch-Controlled Display
Real-time monitoring
Upgradeable platform
Indications for Use
- Improvement of abdominal tone & strengthening of abdominal muscles
- Development of a firmer, more defined abdomen
- Strengthening, toning & firming of buttocks, thighs, calves
- Enhanced muscle tone & firmness in arms (biceps/triceps)
- Support for post-recovery muscle conditioning
Ideal Patient Profile
- Adults seeking healthy weight management & non-invasive sculpting
- No physical diseases interfering with treatment
- No metallic implants or internal metal substances
- Commitment to complementary hydration & active lifestyle
Advanced Engineering & Smart Interface
HI-EMT Technology
High-energy focused electromagnetic waves trigger intense muscle contractions (supramaximal) — far beyond voluntary action — leading to muscle fiber hypertrophy, deeper structural reshaping, and significant fat breakdown processed naturally over weeks.
Smart Touch Control
Fully responsive 8-inch touch display with adjustable brightness. Real-time feedback: remaining time, active channels, treatment mode, intensity level, and attachment status. Guides the clinician step-by-step for error-free protocols.
Real-Time Monitoring & Guidance
During each session, the screen tracks treatment progress, frequency, energy stages, and session duration. Ensures maximum safety and allows fine-tuning according to patient tolerance.
Operating Environment & Safety
Regular disinfection of device and accessories prevents cross-contamination.
Controlled environment (10°C–40°C, RH ≤93% storage) ensures performance.
Ensures precise placement of handpieces.
Fire extinguisher, first aid ready, away from EMI sources.
Contraindications & Safety
Contraindications
- Active skin wounds or lesions (avoid direct contact)
- Cardiac diseases (arrhythmias) & implanted pacemakers or ICDs
- Cancer patients (active malignancy)
- Infectious diseases / acute illness
- Pregnancy, lactation, or menstruation period
- Post-major surgery / uncontrolled diabetes
- Presence of any metallic implants, IUD, or metal fragments
- Cerebrovascular diseases (stroke history)
Possible Side Effects
- Transient muscle soreness (similar to intense workout)
- Temporary muscle spasms / fasciculations
- Mild joint or tendon discomfort
- Localized redness, skin flushing (resolves rapidly)
- Reduction in adipose tissue (expected outcome)
Device Configuration & Technical Specifications
Model: MYOslim Quad White (E-02 Series)
| Input Power Supply | AC 110V–220V, 50/60Hz |
| Output Maximum Power | 0–6500W (adjustable) |
| Frequency Range | 5 – 200 Hz (dual mode: Mode I 5-60Hz, Mode II 30-100Hz, up to 200Hz) |
| Screen Size | 8.0 inches high-brightness touch display |
| Machine Color | Pearl White / Silver accents |
| Handles Included | 2 high-intensity magnetic handpieces (interchangeable, calibration required after exchange) |
| Accessories | Protective cushions, adjustable belts (38/80/90/100cm), power cable, electronic manual |
| Fuse Protection | Replaceable fuse at rear panel, spare included |
Therapy Protocol & Preparation
Pre-Treatment Checklist
- Communicate treatment plan and realistic expectations
- Ensure patient is well-hydrated and healthy
- Wear appropriate, loose clothing (no metal fasteners)
- Avoid heavy meals 1 hour before session
- No alcohol consumption prior to treatment
- Remove all metallic jewelry and accessories
Handling & Operation
- Attach belt to fix handle to target area (abdomen, buttocks, arms, thighs, calves)
- Touch probe vertically to skin, then power ON and adjust intensity gradually
- Real-time parameters: time (adjustable 0–30min typical), frequency stages
- Press start/pause for patient comfort; resume as needed
- After session: post-treatment hydration, avoid intense heat exposure
Maintenance, Cleaning & Warranty
Device Care
• Housing: wipe with damp cloth, mild soapy water. Do not use alcohol-based solvents on painted surfaces.
• Handles & accessories: clean with 75% alcohol before and after each patient. Do not fully immerse.
• Fuse replacement: turn off power, unplug, then replace fuse located near handle junction.
Warranty & Service
Main unit warranty: 1 year (from purchase date) covering manufacturing defects.
Accessories warranty: 3 months (belts, handles, cushions).
Extended support available; do not disassemble without authorization. Contact authorized distributor for service.
Electromagnetic Compatibility & Compliance
Radiated Electromagnetic Field Immunity: Tested according to IEC 61000-4-3:2020, performance level 30V/m with 80% AM modulation. The device is designed for professional healthcare environments; maintain separation from high-frequency surgical equipment or portable RF communications.
EMC compliant Overcurrent protection Stable output across 110-220V
For technical support or service requests, refer to authorized distributor channels.
Troubleshooting Quick Guide
| Fault Phenomenon | Possible Cause | Remedy |
| Device not powering on | Power cord loose / switch off / fuse blown | Check socket & switch; replace fuse |
| Handle not working | Loose connection / handle not calibrated after exchange | Reinsert handle firmly; recalibrate after power off/on |
| Screen unresponsive | Interference or temporary glitch | Power cycle machine, avoid EMI sources |
| No muscle contraction sensation | Low intensity setting / improper belt placement | Increase intensity gradually; ensure skin contact and belt tension |
Federal or local regulations may apply. This device should be operated exclusively by trained professionals. Do not use in presence of flammable anesthetics or oxygen-rich environments. The safety and efficacy depend on correct patient selection and operation according to the official user manual.
For complete instructions, refer to the digital manual included with each MYOslim Quad White system. All specifications subject to manufacturer’s continuous improvement.
| Weight | 50 kg |
|---|---|
| Dimensions | 56 × 53 × 40 cm |
| Technology | EMS,HIEMT,RF |
| Primary Function | Non-invasive body sculpting, muscle toning, strengthening, and fat reduction |
| Indications | Abdomen, buttocks, thighs, calves, arms |
| Frequency Range | 5 – 200 Hz |
| Output Maximum Power | 0 – 6500 W |
| Input Power Supply | AC 110V – 220V, 50/60 Hz |
| Screen | 8.0 inch touch-controlled display, brightness adjustable |
| Machine Color | Pearl White |
-
Description
Medical Aesthetics · HI-EMT Series
MYOslim Quad White
Advanced High-Intensity Focused Electromagnetic Technology | Non-Invasive Body Sculpting & Muscle ConditioningEMS Body Sculpt series integrates HI-EMT Technology (High‑Intensity Focused Electromagnetic Waves) to induce supramaximal muscle contractions — stimulating deep muscle growth, remodeling internal structure, and promoting simultaneous fatty acid release from triglycerides. The result: visible toning, firming, and reshaping without surgery.
5–200Hz frequency range
8″ Touch-Controlled Display
Real-time monitoring
Upgradeable platformIndications for Use
- Improvement of abdominal tone & strengthening of abdominal muscles
- Development of a firmer, more defined abdomen
- Strengthening, toning & firming of buttocks, thighs, calves
- Enhanced muscle tone & firmness in arms (biceps/triceps)
- Support for post-recovery muscle conditioning
Ideal Patient Profile
- Adults seeking healthy weight management & non-invasive sculpting
- No physical diseases interfering with treatment
- No metallic implants or internal metal substances
- Commitment to complementary hydration & active lifestyle
Note: Suitable only for adults. Pre-treatment consultation required.Advanced Engineering & Smart Interface
HI-EMT Technology
High-energy focused electromagnetic waves trigger intense muscle contractions (supramaximal) — far beyond voluntary action — leading to muscle fiber hypertrophy, deeper structural reshaping, and significant fat breakdown processed naturally over weeks.
Smart Touch Control
Fully responsive 8-inch touch display with adjustable brightness. Real-time feedback: remaining time, active channels, treatment mode, intensity level, and attachment status. Guides the clinician step-by-step for error-free protocols.
Real-Time Monitoring & Guidance
During each session, the screen tracks treatment progress, frequency, energy stages, and session duration. Ensures maximum safety and allows fine-tuning according to patient tolerance.
Operating Environment & Safety
Clean & Hygienic
Regular disinfection of device and accessories prevents cross-contamination.Temperature/Humidity
Controlled environment (10°C–40°C, RH ≤93% storage) ensures performance.Adequate Lighting
Ensures precise placement of handpieces.Ventilation & Safety
Fire extinguisher, first aid ready, away from EMI sources.Contraindications & Safety
Contraindications
- Active skin wounds or lesions (avoid direct contact)
- Cardiac diseases (arrhythmias) & implanted pacemakers or ICDs
- Cancer patients (active malignancy)
- Infectious diseases / acute illness
- Pregnancy, lactation, or menstruation period
- Post-major surgery / uncontrolled diabetes
- Presence of any metallic implants, IUD, or metal fragments
- Cerebrovascular diseases (stroke history)
Possible Side Effects
- Transient muscle soreness (similar to intense workout)
- Temporary muscle spasms / fasciculations
- Mild joint or tendon discomfort
- Localized redness, skin flushing (resolves rapidly)
- Reduction in adipose tissue (expected outcome)
Important: Avoid heavy meals 1 hour before and after treatment. Remove metal jewelry before use. Always start intensity at lowest level and increase gradually based on patient tolerance.Device Configuration & Technical Specifications
Model: MYOslim Quad White (E-02 Series)
Input Power Supply AC 110V–220V, 50/60Hz Output Maximum Power 0–6500W (adjustable) Frequency Range 5 – 200 Hz (dual mode: Mode I 5-60Hz, Mode II 30-100Hz, up to 200Hz) Screen Size 8.0 inches high-brightness touch display Machine Color Pearl White / Silver accents Handles Included 2 high-intensity magnetic handpieces (interchangeable, calibration required after exchange) Accessories Protective cushions, adjustable belts (38/80/90/100cm), power cable, electronic manual Fuse Protection Replaceable fuse at rear panel, spare included Therapy Modes: Mode 1 (5–60Hz) – foundational muscle conditioning & toning. Mode 2 (30–100Hz) – high-frequency intensive sculpting & deep tissue remodeling. Frequency & intensity can be adjusted per patient during live session.Therapy Protocol & Preparation
Pre-Treatment Checklist
- Communicate treatment plan and realistic expectations
- Ensure patient is well-hydrated and healthy
- Wear appropriate, loose clothing (no metal fasteners)
- Avoid heavy meals 1 hour before session
- No alcohol consumption prior to treatment
- Remove all metallic jewelry and accessories
Handling & Operation
- Attach belt to fix handle to target area (abdomen, buttocks, arms, thighs, calves)
- Touch probe vertically to skin, then power ON and adjust intensity gradually
- Real-time parameters: time (adjustable 0–30min typical), frequency stages
- Press start/pause for patient comfort; resume as needed
- After session: post-treatment hydration, avoid intense heat exposure
Maintenance, Cleaning & Warranty
Device Care
• Housing: wipe with damp cloth, mild soapy water. Do not use alcohol-based solvents on painted surfaces.
• Handles & accessories: clean with 75% alcohol before and after each patient. Do not fully immerse.
• Fuse replacement: turn off power, unplug, then replace fuse located near handle junction.Warranty & Service
Main unit warranty: 1 year (from purchase date) covering manufacturing defects.
Accessories warranty: 3 months (belts, handles, cushions).
Extended support available; do not disassemble without authorization. Contact authorized distributor for service.Transport & Storage: Store in original packaging, well-ventilated room, temperature -10°C to 40°C, humidity ≤93%. Avoid corrosive gases, shocks during transit.Electromagnetic Compatibility & Compliance
Radiated Electromagnetic Field Immunity: Tested according to IEC 61000-4-3:2020, performance level 30V/m with 80% AM modulation. The device is designed for professional healthcare environments; maintain separation from high-frequency surgical equipment or portable RF communications.
EMC compliant Overcurrent protection Stable output across 110-220V
Manufacturer: Guangzhou Xinwei Electronic Technology Co., Ltd.
For technical support or service requests, refer to authorized distributor channels.Troubleshooting Quick Guide
Fault Phenomenon Possible Cause Remedy Device not powering on Power cord loose / switch off / fuse blown Check socket & switch; replace fuse Handle not working Loose connection / handle not calibrated after exchange Reinsert handle firmly; recalibrate after power off/on Screen unresponsive Interference or temporary glitch Power cycle machine, avoid EMI sources No muscle contraction sensation Low intensity setting / improper belt placement Increase intensity gradually; ensure skin contact and belt tension
Federal or local regulations may apply. This device should be operated exclusively by trained professionals. Do not use in presence of flammable anesthetics or oxygen-rich environments. The safety and efficacy depend on correct patient selection and operation according to the official user manual.
For complete instructions, refer to the digital manual included with each MYOslim Quad White system. All specifications subject to manufacturer’s continuous improvement.
-
Additional information
Weight 50 kg Dimensions 56 × 53 × 40 cm Technology EMS,HIEMT,RF
Primary Function Non-invasive body sculpting, muscle toning, strengthening, and fat reduction
Indications Abdomen, buttocks, thighs, calves, arms
Frequency Range 5 – 200 Hz
Output Maximum Power 0 – 6500 W
Input Power Supply AC 110V – 220V, 50/60 Hz
Screen 8.0 inch touch-controlled display, brightness adjustable
Machine Color Pearl White
-
Reviews (0)
Please use the review form above.
Related Products







Reviews
There are no reviews yet.